Projects

Protein Trafficking and Long-term Potentiation
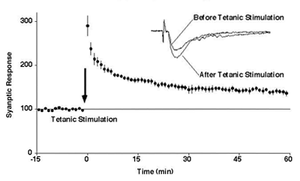
Long-term potentiation (LTP), a persistent enhancement of synaptic transmission following strong neuronal activation, is widely regarded as a cellular substrate of learning and memory. Despite decades of investigation, the molecular processes that sustain this long-lasting increase in synaptic efficacy have remained incompletely understood.
We proposed that LTP is mediated, at least in part, by the activity-dependent recruitment of AMPA-type glutamate receptors to synapses, thereby increasing postsynaptic responsiveness to glutamate. Using a combination of optical imaging and electrophysiological approaches, we demonstrated that LTP induction drives the delivery of new AMPA receptor molecules to synaptic sites[1][2][3]. This process requires Ca2+/calmodulin-dependent protein kinase II (CaMKII) and interactions with PDZ domain–containing proteins.
Importantly, synaptic trafficking during LTP is not limited to AMPA receptors. Multiple synaptic proteins are delivered to postsynaptic sites in a temporally coordinated manner[4], suggesting that LTP involves a broader reorganization of the synaptic proteome.
Notably, this process does not depend on phosphorylation of the AMPA receptor itself[2]. In contrast to earlier models, the fraction of phosphorylated AMPA receptors appears insufficient to account for the magnitude of synaptic potentiation[5]. These findings raise fundamental questions regarding the upstream triggers of receptor trafficking and the mechanisms that stabilize receptors at potentiated synapses.

Liquid-liquid phase separation and synaptic plasticity
Ca2+/calmodulin-dependent protein kinase II (CaMKII) is a central regulator of synaptic plasticity[7]. It is highly enriched in the postsynaptic density (PSD), at levels comparable to structural proteins, a feature that is difficult to reconcile with its canonical role as a catalytic enzyme. In addition, CaMKII forms a unique dodecameric assembly, the functional significance of which has remained elusive.
We identified that Tiam1, a guanine nucleotide exchange factor for Rac, forms a stable complex with CaMKII[8], and further found that CaMKII interacts with and cross-links multiple synaptic proteins. These observations led us to propose that CaMKII undergoes liquid–liquid phase separation, generating condensed protein assemblies within the synapse.
Consistent with this model, inactive CaMKII remains diffusely distributed, whereas Ca2+-dependent activation induces its transition into a condensed phase. Moreover, activated CaMKII promotes the co-condensation of interacting proteins[6]. Such phase-separated assemblies provide a plausible physical mechanism for the rapid accumulation and spatial organization of synaptic proteins during LTP[9].
This framework suggests that synaptic plasticity is not solely governed by biochemical signaling, but also by emergent physicochemical processes that organize synaptic components at the nanoscale.
Molecular Mechanisms of Structural Plasticity of Dendritic Spines
Synaptic potentiation is accompanied by structural remodeling of dendritic spines, the postsynaptic sites of excitatory synapses[4][10][11] (Movie 1). These morphological changes are closely linked to functional plasticity.
Using FRET-based probes, we demonstrated that actin polymerization is rapidly enhanced following LTP-inducing stimulation, shifting the balance toward filament assembly[12][13]. This reorganization persists for tens of minutes, providing a structural basis for sustained synaptic changes.
We further showed that CaMKII directly binds and bundles F-actin in a stoichiometric manner[14]. Upon activation, CaMKII dissociates from F-actin, permitting dynamic remodeling of the actin cytoskeleton during LTP[15]. These findings position CaMKII as a central integrator of signaling and structural processes within the synapse[7][16].


Dynamic Behavior of Cellular Memory Engram
At the systems level, hippocampal place cells encode spatial information by firing selectively when an animal occupies a specific location. These neurons are thought to contribute to the formation of memory engrams; however, their formation, stability, and long-term dynamics remain incompletely understood.
To address these questions, we developed a virtual reality system that enables mice to navigate controlled environments while hippocampal activity is recorded using two-photon Ca2+ imaging[17][18][19]. Ca2+ transients are automatically detected and converted into inferred spike patterns.
This approach enables us to examine how neuronal ensembles evolve over time and how memory representations are stabilized, reorganized, or lost. Through this multi-scale framework, we aim to bridge molecular mechanisms of synaptic plasticity with the emergent dynamics of memory engrams.
References
- ↑ 1.0 1.1
Shi, S.H., Hayashi, Y., Petralia, R.S., Zaman, S.H., Wenthold, R.J., Svoboda, K., & Malinow, R. (1999).
Rapid spine delivery and redistribution of AMPA receptors after synaptic NMDA receptor activation. Science (New York, N.Y.), 284(5421), 1811-6. [PubMed:10364548] [WorldCat] [DOI] - ↑ 2.0 2.1
Hayashi, Y., Shi, S.H., Esteban, J.A., Piccini, A., Poncer, J.C., & Malinow, R. (2000).
Driving AMPA receptors into synapses by LTP and CaMKII: requirement for GluR1 and PDZ domain interaction. Science (New York, N.Y.), 287(5461), 2262-7. [PubMed:10731148] [WorldCat] [DOI] - ↑
Shi, S., Hayashi, Y., Esteban, J.A., & Malinow, R. (2001).
Subunit-specific rules governing AMPA receptor trafficking to synapses in hippocampal pyramidal neurons. Cell, 105(3), 331-43. [PubMed:11348590] [WorldCat] [DOI] - ↑ 4.0 4.1 4.2
Bosch, M., Castro, J., Saneyoshi, T., Matsuno, H., Sur, M., & Hayashi, Y. (2014).
Structural and molecular remodeling of dendritic spine substructures during long-term potentiation. Neuron, 82(2), 444-59. [PubMed:24742465] [PMC] [WorldCat] [DOI] - ↑
Hosokawa, T., Mitsushima, D., Kaneko, R., & Hayashi, Y. (2015).
Stoichiometry and phosphoisotypes of hippocampal AMPA-type glutamate receptor phosphorylation. Neuron, 85(1), 60-67. [PubMed:25533481] [PMC] [WorldCat] [DOI] - ↑ 6.0 6.1
Hosokawa, T., Liu, P.W., Cai, Q., Ferreira, J.S., Levet, F., Butler, C., ..., & Hayashi, Y. (2021).
CaMKII activation persistently segregates postsynaptic proteins via liquid phase separation. Nature neuroscience, 24(6), 777-785. [PubMed:33927400] [WorldCat] [DOI] - ↑ 7.0 7.1
Kim, K., Saneyoshi, T., Hosokawa, T., Okamoto, K., & Hayashi, Y. (2016).
Interplay of enzymatic and structural functions of CaMKII in long-term potentiation. Journal of neurochemistry, 139(6), 959-972. [PubMed:27207106] [WorldCat] [DOI] - ↑
Saneyoshi, T., Matsuno, H., Suzuki, A., Murakoshi, H., Hedrick, N.G., Agnello, E., ..., & Hayashi, Y. (2019).
Reciprocal Activation within a Kinase-Effector Complex Underlying Persistence of Structural LTP. Neuron, 102(6), 1199-1210.e6. [PubMed:31078368] [PMC] [WorldCat] [DOI] - ↑
Liu, P.W., Hosokawa, T., & Hayashi, Y. (2021).
Regulation of synaptic nanodomain by liquid-liquid phase separation: A novel mechanism of synaptic plasticity. Current opinion in neurobiology, 69, 84-92. [PubMed:33752045] [WorldCat] [DOI] - ↑
Hayashi, Y., & Majewska, A.K. (2005).
Dendritic spine geometry: functional implication and regulation. Neuron, 46(4), 529-32. [PubMed:15944122] [WorldCat] [DOI] - ↑
Bosch, M., & Hayashi, Y. (2012).
Structural plasticity of dendritic spines. Current opinion in neurobiology, 22(3), 383-8. [PubMed:21963169] [PMC] [WorldCat] [DOI] - ↑ 12.0 12.1
Okamoto, K., Nagai, T., Miyawaki, A., & Hayashi, Y. (2004).
Rapid and persistent modulation of actin dynamics regulates postsynaptic reorganization underlying bidirectional plasticity. Nature neuroscience, 7(10), 1104-12. [PubMed:15361876] [WorldCat] [DOI] - ↑
Okamoto, K., & Hayashi, Y. (2006).
Visualization of F-actin and G-actin equilibrium using fluorescence resonance energy transfer (FRET) in cultured cells and neurons in slices. Nature protocols, 1(2), 911-9. [PubMed:17406324] [WorldCat] [DOI] - ↑
Okamoto, K., Narayanan, R., Lee, S.H., Murata, K., & Hayashi, Y. (2007).
The role of CaMKII as an F-actin-bundling protein crucial for maintenance of dendritic spine structure. Proceedings of the National Academy of Sciences of the United States of America, 104(15), 6418-23. [PubMed:17404223] [PMC] [WorldCat] [DOI] - ↑ 15.0 15.1
Kim, K., Lakhanpal, G., Lu, H.E., Khan, M., Suzuki, A., Hayashi, M.K., ..., & Okamoto, K. (2015).
A Temporary Gating of Actin Remodeling during Synaptic Plasticity Consists of the Interplay between the Kinase and Structural Functions of CaMKII. Neuron, 87(4), 813-26. [PubMed:26291163] [PMC] [WorldCat] [DOI] - ↑
Okamoto, K., Bosch, M., & Hayashi, Y. (2009).
The roles of CaMKII and F-actin in the structural plasticity of dendritic spines: a potential molecular identity of a synaptic tag? Physiology (Bethesda, Md.), 24, 357-66. [PubMed:19996366] [WorldCat] [DOI] - ↑
Sato, M., Kawano, M., Ohkura, M., Gengyo-Ando, K., Nakai, J., & Hayashi, Y. (2015).
Generation and Imaging of Transgenic Mice that Express G-CaMP7 under a Tetracycline Response Element. PloS one, 10(5), e0125354. [PubMed:25946002] [PMC] [WorldCat] [DOI] - ↑ 18.0 18.1
Sato, M., Kawano, M., Mizuta, K., Islam, T., Lee, M.G., & Hayashi, Y. (2017).
Hippocampus-Dependent Goal Localization by Head-Fixed Mice in Virtual Reality. eNeuro, 4(3). [PubMed:28484738] [PMC] [WorldCat] [DOI] - ↑
Sato, M., Mizuta, K., Islam, T., Kawano, M., Sekine, Y., Takekawa, T., ..., & Hayashi, Y. (2020).
Distinct Mechanisms of Over-Representation of Landmarks and Rewards in the Hippocampus. Cell reports, 32(1), 107864. [PubMed:32640229] [PMC] [WorldCat] [DOI]